24/08/2021
Introduction
18 months on from the world’s fearful response to the arrival of SARS-CoV-2, we provide an alternative to New Zealand’s elimination strategy to one of ‘living with covid-19’. We are now back in level four lockdown indefinitely with escalating PCR positive ‘cases’. We urgently need to reassess New Zealand’s elimination strategy and whether it makes sense given the new information.
The revised strategy takes account of five major developments over the period:
- The infection is far less threatening than originally forecast by authorities, including New Zealand, when they proposed lockdowns and other restrictions. Data from the WHO, CDC and other peer-reviewed studies show the median infection fatality ratio (IFR) is ~0.23%, not the projected 3.6%. The condition is therefore more akin to pandemics in 1957 and 1967 than influenza in 1918. Asymptomatic individuals do not spread the infection, removing the key idea underpinning lockdowns. Long-term health effects (“long covid”) have not proven any different to or more prevalent that those experienced in the recovery period from existing circulating pathogens.
- Questions still remain about the accuracy of the polymerase chain reaction (PCR) test used to diagnose ‘covid-19 cases’. The virus remains yet to be isolated, the sequence of the virus was generated in silico (stitched together from computer databases) and many people who test positive are asymptomatic. In addition, the clinical symptoms associated with covid-19 are not unique.
- It is clear that the average age of death with covid-19 is about the same as our life expectancy (~82 years). Older people are much more likely to die of covid than younger ones.
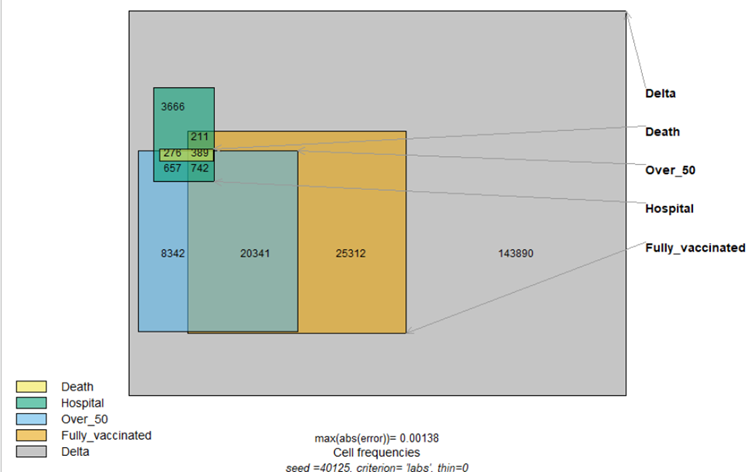
- Very rapid development of vaccines and dissemination of these in New Zealand. The vaccines show some evidence of reducing PCR positive cases, but not of prolonging overall survival or reducing transmission. In many countries now with highly vaccinated populations, there are increasing numbers of breakthrough cases. It is now obvious that vaccines will not stop the spread of the condition long term. In addition, clear evidence shows a major increase in post-vaccination deaths and serious injuries.
- Early treatment protocols are showing promise in the early treatment of cases otherwise destined to be hospitalised.
- New Zealand’s very low incidence of covid-19, with the apparent absence of community transmission for many months, whereas covid-19 cases occur freely throughout the rest of the world. Now, we are faced with yet another lockdown and an increase in case numbers.
The vaunted elimination objective makes re-engagement impossible without an improved vaccine administered as often as necessary to most of the population.
New Zealand cannot sustain economically or socially the years of border closure, threat of lockdowns, social disruption and government debt, needed to reach this position, if it can be reached at all. We believe, frankly, this to be a utopian pipe dream, but necessitating dystopian government dictates. The fabric of our society will be rent – then restitched to what?
We propose an approach that slowly and carefully manages our entry back into a world where covid-19 exists, and where it can exist in New Zealand without causing unacceptable harm.
Guiding Principles
The risks of mortality following covid-19 infection have been grossly exaggerated. As observed in other pandemics, a high degree of ascertainment bias has occurred that has further exaggerated the importance of this condition in the minds of scientists, decision makers and politicians. This has led to an over prioritisation of the illness above many other health issues. In turn, this exaggerated threat has led to mortality and morbidity from other diseases due to the imposition of lockdowns and disruption of usual medical care.
The economic effects of lockdowns and border closures, leading to unemployment and poverty will lead to further health deterioration that is out of proportion to the threat of covid-19. Consistent evidence also highlights that lockdowns do not limit the spread of infection.
Now, it is important to note that hospital treatment for covid-19 patients has improved considerably during the course of the pandemic and that hospital mortality has declined. Potential treatments are also available to reduce morbidity and mortality include the use of both the micronutrient vitamin D and anti-parasitic and anti-viral drug ivermectin. It is also clear that metabolic disease is an important contributor to death with covid-19, and it also raises risk of death from other diseases. Addressing dietary risks related to metabolic disease is also worthwhile to reduce potential harm from covid-19, such as reducing sugar intake.
These guidelines were inspired from those produced by the group who published pandata.org.
Ongoing pursuit of elimination is risky
New Zealand is the only country in the world now continuing to attempt to eliminate cases. Many countries that were attempting to eliminate covid-19 have now given up, such as Singapore, UK and Australia. It is a dead-end strategy which will leave New Zealand isolated and vulnerable, in a (possibly) covid-free bubble. Even if elimination is possible and the reward warrants the financial and social cost, cases will still exist throughout the rest of the world – endemic for the foreseeable future (hundreds of years). To keep it out, New Zealand will need to retain covid-19 border testing indefinitely. Similarly, lockdowns and tracing and testing have no time limit.
There are three ends to the elimination strategy:
- A cataclysmic failure at the border, such as the beginnings of which we are now seeing, or a winter-resurgence within the country, in which infection sweeps quickly through the population. Lockdowns would, like the US and UK, not protect us.
- The infection becomes endemic with low levels of circulation and winter peaks, like the varieties of influenza and coronaviruses that circulate. This is likely to take many years. New Zealand would need to decide a point at which it could open.
- Future vaccines may be developed to completely interrupt transmission of covid-19. The development of the currently available partially effective vaccines has been the quickest ever, and faster than we imagined. We do not yet have evidence that the current vaccines reduce viral transmission through a population. Given performance to date, this evidence might one day eventuate. But the rest of the world is not trying to eliminate covid-19 and appears satisfied with the imperfect protection of the current vaccines. That makes it uncertain whether there will be a commercial incentive to ever invent such a comprehensively protective vaccine, since the existing ones are not as effective as required to maintain population elimination.
Our belief is that none of these exits from the elimination strategy are palatable.
Instead, New Zealand should prepare for, and carefully manage, the inevitable introduction of covid-19 to New Zealand.
Frequently asked questions
Do new variants and strains (lineage B 1.1.7 or delta) pose an increased risk of harm?
Every virus is thought to have thousands of variants. There are over 100,000 alleged variants for covid-19. The fact that there are new strains is not important. What’s important is their effect. With the UK strain, the claim is it transmits easier. We haven’t yet seen any convincing evidence that new strains are more dangerous.
Has there been increased overall mortality as a result of covid-19?
Yes, there has been increased overall mortality in some countries, but not all. Many countries, such as Malaysia, Cyprus, Costa Rica, Uruguay, Japan, Singapore, Denmark, Finland, Ireland, Luxembourg and Malta have not. Excess death is also statistically associated with the period after lockdowns in between country comparisons and between US states. Since the average age of death is close to our life expectancy in almost every country, much of the excess mortality is likely to be related to displaced mortality, and light influenza seasons in recent years, leaving a high number of people who are frail and elderly. It is also clear that some of the excess mortality was due to responses to covid-19, such as abandoning non-invasive ventilation for intubation and mechanical ventilation and prematurely sending infectious patients from hospital to rest homes. Hospital mortality in New York has now dropped by 70% since the beginning of the pandemic.
Does evidence support the wearing of masks to prevent infection?
The best evidence from a randomised controlled trial, the Danish mask study, couldn’t find any evidence to support mask use, particularly cloth ones, to protect the wearer. That also indicates that they are not preventing transmission. And asymptomatic people are unlikely to transmit the infection anyway.
What is the extent of the economic recession?
Globally, the World Bank is saying we are now facing the greatest recession since World War 2, demand in food banks in New Zealand has doubled or trebled and we have now thrown more than 50,000 adults in New Zealand into the dole queue, since March, when lockdowns and border closures began.
The health effects from the widespread panic over covid-19 has also produced many mental problems. For example, there has been an increase in children hospitalised for eating disorders both here in Auckland and in Melbourne. In the UK, mental health scores have deteriorated.
Does asymptomatic spread occur?
A mass testing study in Wuhan, a city of 10 million residents, identified 300 asymptomatic cases, with no evidence of spread of infection from them.
Are you just scientific outliers?
We might seem a minority in New Zealand, but our approach is the same as the Great Barrington Declaration, a view on covid-19 signed internationally by 15,000 medical and public health scientists and almost 44,000 medical practitioners. The counter viewpoint signed by supporters of lockdowns only mustered ~4,200 signatories.
The Plan
Brief guide
- Offer enhanced protection and treatment for covid-19 to vulnerable people.
- End mass testing, contact tracing, quarantine and lockdowns.
- Vaccination should be voluntary and with informed consent and transparency of both efficacy and safety data.
Healthcare recommendations
- Since approximately half of fatalities worldwide with covid-19 have occurred in people living in rest homes, this should be the focus of protection. Effort should be given to protecting those who are at high risk of fatality from covid-19, which are individuals aged greater than seventy-five years, particularly those living in supported residential care, and those with metabolic health conditions, such as diabetes, obesity and cardiovascular disease. Measures to protect these people could include regular testing of health workers with respiratory symptoms, who have a high level of exposure to vulnerable people. Strong exclusion policies for workers with respiratory symptoms are important. Ensure people with covid-19 are not in contact with vulnerable people during their infectious period. Other measures include:
- Minimise the number of nursing home staff a resident is exposed to.
- Provide outdoor areas for socialisation of rest home residents where transmission of the infection is likely to be lower.
- Enforce strict exclusion policies related to workers or visitors with any respiratory symptoms.
- Encourage supplementation of vitamin D and sun exposure for vulnerable people, since trial evidence supports the use of this micronutrient to prevent intensive care admission in hospitalized patients.
- End mass testing for the infection and contact tracing. The test should be only used within a clinical context of a characteristic clinical picture, compatible with a lower respiratory infection within hospitalised individuals.
- Increase capacity in hospitals and intensive care units to cope with seasonal demands of respiratory illnesses, including covid-19. As stated, early treatment on diagnosis promises to reduce admissions
- Cases should only include those who test PCR positive, at a limited cycle threshold value, with compatible symptoms of a respiratory infection.
- Deaths from covid-19 should include only those who fulfil the criteria of being an active covid-19 case temporally related to their death, with no other likely competing cause.
- Eliminate mask wearing in the community, since evidence does not support their use to prevent infection in the community.
- Vaccination should be entirely voluntary with informed consent of the risks and benefits as more information about their efficacy and side effects come to hand. Vaccination for children of school age should be withdrawn since they are not at appreciable risk of covid fatality. Dangers of exposure to the vaccine, particularly to pregnant women, should be made clear and Ministry of Health information updated accordingly. Vaccination passports or any form of discrimination based on vaccination status should be abandoned, since the vaccines do not convincingly reduce SARS-CoV-2 transmission.
- Consider the routine use of vitamin D and ivermectin in the treatment of hospitalised covid-19 infection.
Societal recommendations
- Abandon the use of either regional or national lockdowns to contain viral spread, since they are unnecessary, economically disastrous and ineffective.
- Schools, childcare centres and universities should not be subject to restrictions and face-to-face learning should have no restriction since children are at extremely low risk for covid fatality.
- End all restrictions on businesses.
- Undergo a phased re-introduction of normal travel across New Zealand’s border. At first, a risk-based approach may be undertaken, as shown in the following web app and accompanying paper, which has been published in the New Zealand Medical Journal. This strategy indicates a method for opening NZ’s border, based on the estimated prevalence of covid-19 infection in the country of the traveller’s origin. This would enable travellers to come from several countries immediately who have a very low prevalence of covid-19. New Zealand should then aim to end travel restrictions completely, should this initial strategy be successfully enacted. In support of such a stance, the European CDC, for example, has now recommended the dropping of covid-19 testing and quarantine across borders.
- End the covid-19 elimination strategy in New Zealand. With cases widespread globally, it is clear that such a strategy is neither sustainable nor beneficial from a perspective which considers both the costs and benefits of such a strategy to New Zealand. Eventually, infection is likely to become endemic and part of the usual seasonal respiratory illnesses that affect New Zealanders every year.